Specific Heat Formula: A Complete and Easy Guide for Students
Specific Heat FormulaUnderstanding the specific heat formula is an important step in learning basic science. Heat and temperature are part of our daily lives. We feel heat from the sun, we cook food using heat, and we warm our homes during winter. But have you ever wondered why some materials heat up quickly while others take longer? The answer lies in the specific heat formula.
In simple terms, the specific heat formula helps us measure how much heat energy is needed to change the temperature of a substance. Scientists, engineers, and students use this formula to understand how heat moves and how different materials react to heat.
This article will explain the specific heat formula in a simple and clear way. You will learn what specific heat is, how the formula works, where it is used, and why it is important in everyday life.
Understanding the Idea Behind the Specific Heat Formula
Before learning the specific heat formula, it is important to understand what specific heat means.
Specific heat is the amount of heat energy required to raise the temperature of one unit mass of a substance by one degree.
In other words, it tells us how much heat a material needs to warm up.
For example:
- Water needs a lot of heat to increase its temperature.
- Metal heats up much faster with less heat.
This means water has a high specific heat, while many metals have a low specific heat.
The specific heat formula helps us calculate this heat energy.
The Specific Heat Formula Explained Simply
The specific heat formula is written like this:
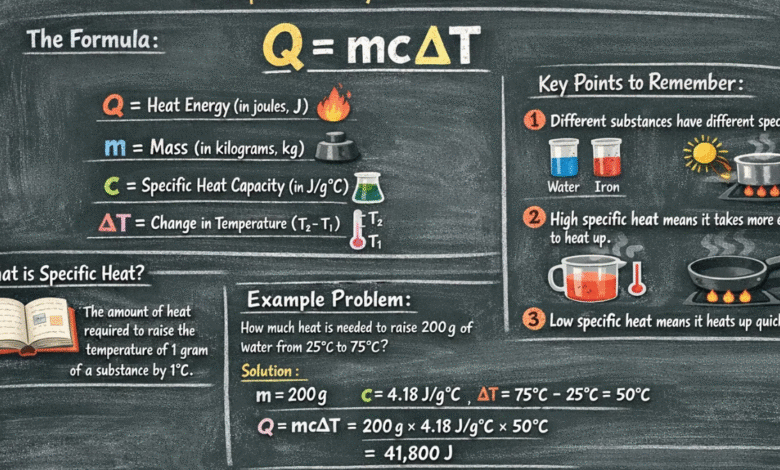
Q = m × c × ΔT
Let’s break it down so it becomes easy to understand.
- Q = Heat energy added or removed
- m = Mass of the substance
- c = Specific heat capacity
- ΔT = Change in temperature
The change in temperature (ΔT) means:
Final Temperature – Initial Temperature
So, the formula can also be written as:
Q = m × c × (T₂ − T₁)
This simple formula helps scientists calculate how much heat is needed to heat or cool a material.
Meaning of Each Part of the Specific Heat Formula
To understand the specific heat formula, it helps to know what each symbol means.
| Symbol | Meaning | Description |
|---|---|---|
| Q | Heat Energy | The amount of heat added or removed |
| m | Mass | The amount of material present |
| c | Specific Heat Capacity | Heat required to raise temperature of 1 unit mass |
| ΔT | Temperature Change | Difference between final and initial temperature |
Each part of the specific heat formula works together to calculate heat energy.
If any value changes, the result will change too.
Why the Specific Heat Formula Is Important
The specific heat formula is very useful in science and everyday life.
It helps us understand how heat energy behaves.
Here are some reasons why it is important:
Helps Scientists Study Heat
Scientists use the specific heat formula to study how different materials react to heat.
Important in Engineering
Engineers use this formula to design:
- Engines
- Heating systems
- Cooling systems
- Machines
Useful in Weather and Climate Studies
Water has a high specific heat, which helps control Earth’s climate.
Oceans absorb heat slowly and release it slowly. This helps keep temperatures stable.
Helps in Cooking
Cooking also involves heat transfer. Knowing how heat works helps chefs cook food evenly.
Real-Life Examples of the Specific Heat Formula
The specific heat formula is not just a classroom idea. It appears in many real-life situations.
Heating Water
Water takes longer to heat compared to metal because its specific heat is high.
That’s why:
- Pots heat quickly
- Water inside the pot heats slowly
Car Engines
Car engines produce a lot of heat.
Coolants like water absorb this heat using the specific heat formula principle.
Climate Near Oceans
Cities near oceans have milder climates.
Why?
Because water absorbs and releases heat slowly due to its high specific heat.
Solar Energy Systems
Solar heaters use materials that store heat effectively. Engineers use the specific heat formula to choose these materials.
Units Used in the Specific Heat Formula
Units are very important when using the specific heat formula.
Different systems of measurement may be used.
Common units include:
| Quantity | Common Unit |
|---|---|
| Heat Energy (Q) | Joule (J) |
| Mass (m) | Kilogram (kg) |
| Specific Heat (c) | J/kg°C |
| Temperature Change (ΔT) | °C or K |
When using the specific heat formula, all units must match the same system.

Step-by-Step Example Using the Specific Heat Formula
Let’s solve a simple problem using the specific heat formula.
Problem
How much heat is needed to raise the temperature of 2 kg of water by 5°C?
Specific heat of water = 4200 J/kg°C
Step 1: Write the Formula
Q = m × c × ΔT
Step 2: Insert the Values
Q = 2 × 4200 × 5
Step 3: Multiply
Q = 42000 Joules
Final Answer
42,000 Joules of heat are needed.
This example shows how the specific heat formula helps us calculate heat energy easily.
Specific Heat of Common Materials
Different materials have different specific heat values.
Here are some common examples.
| Material | Specific Heat (J/kg°C) |
|---|---|
| Water | 4200 |
| Aluminum | 900 |
| Copper | 385 |
| Iron | 450 |
| Gold | 129 |
| Silver | 235 |
From this table, we can see that water has one of the highest specific heat values.
This is why water is often used in cooling systems.
Factors That Affect the Specific Heat Formula
The specific heat formula itself stays the same, but some factors affect the results.
Type of Material
Different substances store heat differently.
For example:
- Water stores more heat
- Metals store less heat
Mass of the Object
More mass means more heat is needed.
A large pot of water needs more heat than a small cup.
Temperature Change
If the temperature change is larger, more heat energy is required.
These factors all play a role in the specific heat formula.
Common Mistakes When Using the Specific Heat Formula
Students sometimes make small mistakes while using the specific heat formula.
Here are some common ones.
Mixing Units
Using grams instead of kilograms without conversion can give wrong answers.
Forgetting Temperature Difference
Students sometimes forget to calculate ΔT.
Remember:
ΔT = Final Temperature – Initial Temperature
Using Wrong Specific Heat Value
Each material has its own specific heat.
Using the wrong value can change the answer.
How the Specific Heat Formula Is Used in Science and Technology
The specific heat formula is widely used in many fields.
Mechanical Engineering
Engineers calculate heat transfer in machines.
Environmental Science
Scientists study how oceans store heat using this formula.
Material Science
Researchers use the formula to understand how materials behave under heat.
Energy Systems
Power plants and heating systems rely on heat calculations.
Without the specific heat formula, understanding heat energy would be very difficult.
Easy Tips to Remember the Specific Heat Formula
Here are some simple tips to remember the specific heat formula.
- Think of Q = m × c × ΔT as Heat = Mass × Material × Temperature change
- Always check units
- Calculate temperature change first
- Use the correct specific heat value
These small tips can make solving problems much easier.
Fun Facts About Specific Heat
Learning science becomes more exciting with interesting facts.
- Water’s high specific heat helps life survive on Earth.
- Oceans store huge amounts of heat energy.
- Desert sand heats quickly because it has low specific heat.
- Ice and snow help keep polar regions cold because of heat absorption.
All these examples show the power of the specific heat formula in understanding nature.
Conclusion
The specific heat formula is one of the most important concepts in physics and thermodynamics. It explains how heat energy changes the temperature of different materials.
By using the simple equation Q = m × c × ΔT, scientists and students can calculate the heat needed to warm or cool substances. From cooking food to designing engines and studying climate, the specific heat formula plays a major role in our world.
Understanding this formula also helps us appreciate how materials behave around us. Water cooling a car engine, oceans controlling climate, and metals heating quickly on a stove all follow the same basic principle.
In the end, the specific heat formula is more than just a scientific equation. It is a powerful tool that helps us understand heat, energy, and the way our physical world works.

